Helium leak testing is one of the oldest and most developed members of the family of trace-gas-based leak testing methods. Even though the title contains the word “helium,” this article is intended as an overview of trace gas methods in general.
Trace gas methods measure leaks directly, i.e. they aim to determine the quantity of material escaping from the part under test. This is in contrast with other, non-trace-gas-based methods that measure the effects of the leak (as is the case with pressure-decay leak testing). Because of this, these methods are generally capable of measuring very small leaks and can be used to test large or complex parts because part volume has no direct effect on the reliability of the test.
Why helium?
Helium has been the dominant trace gas of choice for many years. In the recent decades, occasional shortages and rising prices forced the leak testing industry to look for alternatives such as a Hydrogen/Nitrogen mix, SF6 and various refrigerants.
The ideal trace gas should:
- Be inert – it must not react with any of the materials with which it comes into contact
- Have a very low presence in ambient air
- Be widely and reliably available, and
- Be low cost
Out of this list, with the exception of the last two, helium has no match. Its 5ppm ambient presence in atmosphere, inert nature and very small atomic dimensions make it ideally suited for use as a trace gas.
Let’s review commonly used trace-gas based test methods by first looking at how a leak test system is constructed.
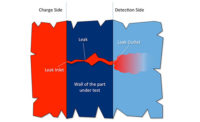
Main Components of a Trace Gas Leak Test System
Trace gas leak testers have two main components:
- A charge system used to manage the pressurization/venting process of the part
- A detection system that must have sufficient sensitivity to detect leaks in the range of interest
Charge Systems
The accuracy of the leak test heavily depends on the concentration and homogeneity of the trace gas inside the part. The most common challenge is the fact that parts usually contain air when they enter the leak test station. If the part is capable of handling vacuum, evacuating this residual air using a dedicated vacuum source (vacuum pump, air-driven vacuum generator, etc.) is the best way of ensuring high trace-gas concentration. When this is not possible, the other option is to allow trace gas to flow through the part (preferably exiting at a point farthest away from the charge port). The latter method can severely increase trace gas consumption and usually is the choice of last resort.
A typical charge sequence consists of the following:
- Evacuation or trace gas flush
- Charge to pressure
- Exhaust (after the test is complete)
- Re-evacuation or air/other inert gas flush
- Venting to atmosphere
Gas management, especially during the exhaust phase, is very important: Keeping high concentrations of trace gas away from the leak detection system is the best way of ensuring the repeatability of test results.
Charge systems can range from a few manual valves on a manifold that is manually connected to the test part through a test line to fully automated valve controls and part connection tooling.
Robust and reliable tooling design is very important, especially in case of the part connection(s). That’s because any leaks will be added to the part leakage and can very easily cause false rejects.
Detection Systems
The most important component of these systems is the leak detector. There are a number of choices—selecting the right one requires careful consideration of the test requirements and the cost. Some of the most commonly used detector types are summarized in Table 2.
The leak detector’s sensitivity determines the leak rate range that can be detected. Depending on the test method, the full range of the leak detector may not equal the detection range of the test system. In most detection systems, the leak detector is sub-sampling gas and some of the trace gas signal is lost. A typical example would be a hard-vacuum leak test system in which the helium mass spectrometer only detects a portion of the helium entering the vacuum chamber.
Take, for example, a larger vacuum chamber such as fuel tank tester. Out of a 10-5 std.cm3/sec leak into the vacuum chamber, only 10-7 std.cm3/sec reaches the mass spectrometer due to the loss of signal through the chamber evacuation pumps.
Other important parts of the detection system are the collection areas (vacuum chamber and pumps, accumulation chamber, etc.), the sampling circuits and any associated equipment providing auxiliary functionality (e.g. carrier gas circuits). These will be detailed in the section about the individual test methods.
Most Common Test Methods
Table 3 summarizes the most common trace-gas-based methods together with their capabilities, the associated system cost and maintenance requirements.
Hard Vacuum Leak Testers
This is the most traditional and most accurate test method. It’s capable of measuring very small leaks, in some cases as low as the 10-9 std.cm3/sec range. The part is placed inside a vacuum chamber which is in turn evacuated using a vacuum pump or pumps. Once the chamber is at a sufficiently low pressure, the mass spectrometer or residual gas analyzer is connected to the chamber. When the trace gas level in the chamber gets low enough for the test to begin, the part is pressurized with trace gas and is held under pressure for the duration of the test. The level of trace gas is continuously monitored in the chamber and if it exceeds a critical limit, the test part is rejected.
This is a very mature technology. There are many safeguards built into the system to detect malfunctions of critical system components. Cycle times can be as short as 20 to 30 seconds, making it the quickest test method available.
All this of course comes at a cost—hard-vacuum systems are usually the most expensive and require the most skilled maintenance. Just like with vehicles, a little regular maintenance goes a long way toward keeping the testers operational. There are many hard-vacuum leak testers out in the field today that are over 30 years old, have been re-tooled multiple times and are still running as reliably as ever.
Why use helium? About 99% of hard vacuum leak testers use helium simply because of the wide availability of helium mass spectrometer leak detectors that have been “industrialized” over the last few decades. Using hydrogen in vacuum is challenging at best because most commonly used metals outgas the large amounts of hydrogen that have been absorbed by them during the manufacturing process. This creates such high levels of unstable background levels that detecting small leaks becomes impossible. Other gases such as refrigerants are used when the leak detector is a residual gas analyzer (RGA). This is a full-blown mass spectrometer capable of analyzing gases in a wide range of atomic mass units (AMUs).
Accumulation Testers
The most common problem with hard-vacuum testers is the need to maintain a high-vacuum system that operates at a very high frequency, cycling between atmospheric pressure and high-vacuum sometimes as often as every 30 seconds.
As its name suggests, the accumulation method measures the accumulated trace gas in a closed volume. To be more precise, it monitors the concentration of trace gas in the test chamber over time. The rate of change of the concentration is directly related to the leak rate: the higher the leak rate, the faster the concentration increases. Some systems calculate the rate of change on very short intervals of time, giving something close to a live leak rate reading. Others wait for the duration of the test and simply look at total increase in concentration.
It is very important to ensure that the system doesn’t lose its sensitivity for one reason or another. To verify test integrity, some systems require the running of a challenge part (a known leaker) periodically. The other option is using a so-called background leak. This is a leak that is constantly connected to the test chamber and has to be detected at the end of the test for the test to be successful.
This background leak is typically 10-20% of the reject level. In addition to being a “sanity check” at the end of the test, it also eliminates the problem of marginal parts. If a part is leaking at 90% of the reject limit, adding a 20% background leak causes the part to be rejected. The level of background should be determined by considering a collection of factors (variation in the parts, uncertainty of the test parameters, etc.).
The air inside the accumulation chamber is usually agitated with fans or by adding an air recirculation loop to the system. The benefit of the recirculation approach is that it creates a closed loop of air flow which the entire chamber atmosphere will be forced to circulate through at some point. By sampling the recirculation loop, one can ensure that all the air in the chamber is sampled.
As with all atmospheric leak test methods, it’s very important to consider the air flow patterns inside the chamber and ensure that there are no “blind spots” or areas where the air flow around the part is limited.
Why use Helium? The simple answer is you don’t have to. After sniffing, accumulation testing was the second method to enter the non-helium camp. Forming gas (a widely available 5% hydrogen/95% nitrogen mix) can be successfully used as a trace gas, provided the background levels are well controlled. If the test environment is kept free of hydrocarbon-emitting sources (e.g. natural gas-powered forklift trucks) forming gas is an excellent alternative.
Carrier Gas Testers
Accumulation testers do not usually provide real-time leak rate information and also suffer from a relatively high level of sensitivity to variation in background levels of trace gas in the atmosphere in and around the accumulation chamber.
Carrier-gas leak testing can instead be used with smaller parts. This method does not accumulate the trace gas in the test volume. Rather, it uses a stream of trace-gas-free gas (e.g. nitrogen) to sweep the chamber. The carrier gas exiting the chamber is sampled by the leak detector and by measuring the concentration of trace gas in the carrier stream, the leak rate of the test part can be calculated.
The same precautions used with accumulation testing (e.g., the addition of a background leak) are highly recommended.
The big advantage of this test over accumulation is twofold: It provides real-time leak information and eliminates the background dependence of accumulation.
The challenge is that the size of the test chamber and the detectable leak rate are linked. The bigger the test chamber, the higher the flow that is needed to ensure that all areas are sampled in a timely manner. A higher carrier gas flow, however, dilutes the trace gas signal, making the measurement more difficult.
This method is usually applied for smaller parts or assemblies.
Why use helium? Again, the answer is it is usually not necessary. This method easily lends itself to using other trace gases provided the leak detector has a high enough sample flow.
Nitrogen Purge Technology
Carrier-gas testing readily lends itself to testing areas of larger parts. The challenge is sealing. The technique assumes a well-sealed test chamber that prevents potentially contaminated air from interfering with the test. Achieving this seal in a production environment is not always practical or even possible.
The nitrogen purge technology method eliminates this problem. By using a trace-gas-free gas as a seal between the test chamber, the isolation of the test chamber from the outside world becomes less of a challenge.
At the beginning of the test, the chamber is purged with a high flow of nitrogen. This forces the atmospheric air out. By maintaining a gas “curtain” around the perimeter of the chamber, the air is kept out for the duration of the test, eliminating any background issues.
Sniffers
Originally, sniffers were accessories to one of the methods described above, used mostly for leak location. In the recent years, with the entire industry become more and more cost-conscious and with the advance of robotics, sniffing as a leak quantifying method has gained ground.
The key to a successful sniffing operation is gas management. All possible trace gas sources in the area must be eliminated and an ample amount of fresh air must be available at all times. Many applications involve a so-called sniffer booth that isolates the test area from the rest of the production floor.
In closing
Just as with medieval rulers, the succession process of helium has been a turbulent affair. As of this writing, a universally acceptable replacement has not been found for trace-gas-based leak testing. Helium’s relatively low ambient presence in atmosphere, its inert nature and its very small atomic dimensions make it ideally suited for use as a trace gas. By using the right recovery methods, helium can still be the best solution despite the occasional difficulties in its procurement.
My hope is that this article has served as a suitable primer for those of you who are new to helium leak testing, to better understand its advantages, the commonly used trace-gas-based test methods, and how best to attain a reliable and repeatable test.