Case Study
Material Properties
Factoring them into simulation leads to higher performance spinal disc implants that deliver long-term pain relief.
Source: MSC Software Corporation

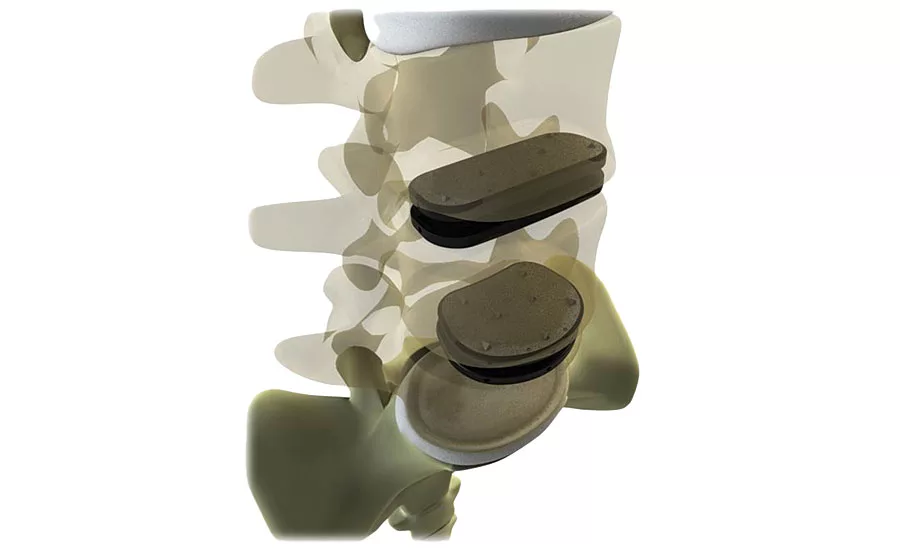
Fig. 1 Simulation software that incorporates material properties data produces more accurate predictions of the composite behavior for materials such the fiber reinforced plastics used to create spinal disc implants such as the one depicted by this simulation model. Source: MSC Software Corporation
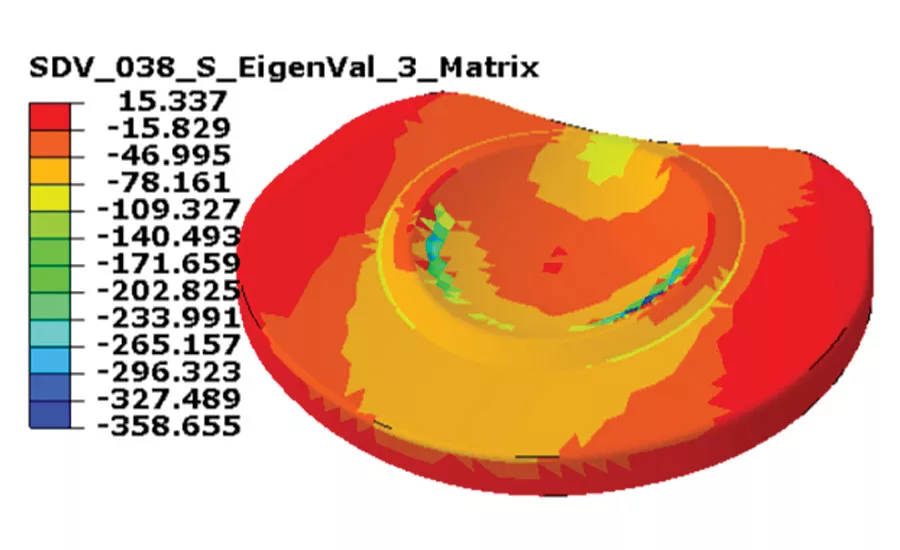
Fig. 2 The contour shows high compressive stresses in the plastic matrix near the inside of the endplate. Source: MSC Software Corporation
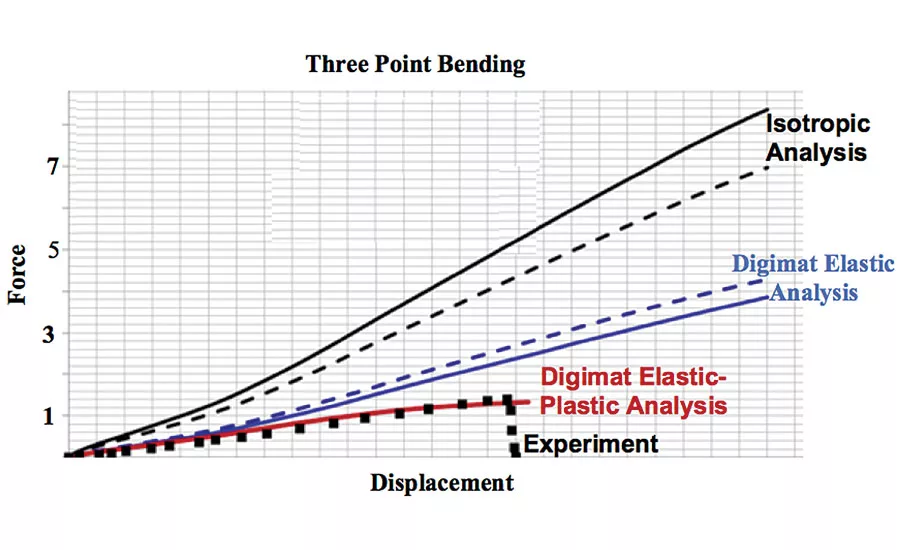
Fig. 3 Digimat material model accounting for manufacturing process results in more accurate simulations that reduce test/analysis iterations and improve performance predictions by more than 170 percent. Source: MSC Software Corporation
One of the most promising surgical options for treating spinal injury and deterioration of spinal discs is the use of artificial discs to replace the patient’s natural spinal disc.
The materials used in these artificial discs are an important factor in the development of this technology. The discs must be made from materials that are safe to be implanted in the human body, do not cause allergic reactions, and are wear resistant and compatible with medical imaging (MRI for example). Fiber-reinforced plastics are used more and more in today’s orthopedic implants because of their resistance to wear and improved mechanical properties.
The challenge when designing implants that take advantage of reinforced plastics is in predicting the as-manufactured material performance. The mechanical properties of an implant designed with fiber reinforced plastics can vary widely depending on the use of the material and how the implant is manufactured. The injection molding process used to manufacture the implant will affect the fiber orientations throughout the part and the induced mechanical properties.
Typical analysis assumes the material is homogeneous and isotropic for simplicity. In reality, fiber alignments continuously change throughout the implant, resulting in a heterogeneous, anisotropic material. Wrongly aligned fibers or not appropriately accounting for the fibers’ effect on material performance can lead to a softer or stiffer implant than designed, and even premature, unpredictable failure.
SOLUTION
Medicrea solved the isotropic versus anisotropic problem by using MSC Software’s Digimat, a non-linear multi-scale material and structure modeling software platform, to create a micromechanical material simulation of the implant design. Unlike a homogeneous isotropic analysis, Digimat’s heterogeneous anisotropic analysis considers the variation in fiber content and alignments and how they affect material properties. Figure 1 depicts how a simulation model shows variations in fiber orientation throughout the plate of a spinal implant.
Medicrea used the software to produce accurate predictions of the composite behavior for materials such as fiber reinforced plastics. Medicrea was able to start with the same finite element model of the implant that was used for the previous analysis because the simulation software works with any major finite element solver.
Next, engineers requested the injection mold simulation results from the manufacturer. This analysis assures that the implant’s mold has been designed properly for manufacture, but the results can also be used in the finite element analysis of the implant itself.
By selecting intelligent material models of the reinforced plastic from the software’s material library and creating models using integrated tools, Medicrea engineers mapped the fiber orientations, residual temperatures and residual stresses onto the structural analysis model. The intelligent material model is a function of fiber orientations instead of an approximate/guessed value, so it adjusts the material stiffness at every location throughout the implant.
With this preliminary work done, engineers were ready to conduct the usual analysis with one key exception: the approximate value for material stiffness will be replaced by the local material model. The simulation software takes care of tying the intelligent material into the analysis solution so that the analyst can focus on designing the implant’s performance, not guessing at which material property might give the best results. Figure 2 depicts the results of a simulation that incorporates material property data and fiber alignment into the analysis.
The attention to material details yields more accurate simulations that reduce test/analysis iterations and improve performance predictions. In the case of the spinal implant design, the original isotropic simulation over-predicted the implant’s stiffness by as much as 170 percent (Figure 3).The same simulation using a material model that accounted for both changes in fiber orientations, as well as plastic deformations, matched the test results almost perfectly. These simulation capabilities promote optimization of production processes that yield mechanically efficient implants that can be implanted safely, perform to or exceed design specifications, and last for years to spare patients repeat surgeries.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!