KMC Launches Medical Device and Instrumentation Life Cycle Support Program
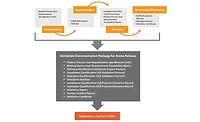
MERRIMACK, NH-Elbit Systems of America’s KMC Systems, provider of contract design, manufacture and maintenance of medical devices and instrumentation, introduces KMC 360, the company’s complete program for contract design, manufacture and maintenance. The program is designed to support OEMs and start-ups throughout the entire medical device and instrumentation product lifecycle by centralizing all outsourcing support. The company’s proven process streamlines communication, strengthens product quality and results in time-to-market advantages.
“Relying on multiple partners throughout a product’s lifecycle can result in market launch delays, increased costs and inefficiencies,” says Patrick W. McNallen, president, KMC Systems. “Just as a circle is all encompassing, so too is KMC Systems’ portfolio of services. Our customers work with one partner that has the relevant experience and skill set to develop, produce and maintain complex medical devices and instrumentation.”
KMC 360 helps medical companies bring products to market on time and on budget as well as reduce manufacturing and field service costs, which is especially critical in times of economic difficulty. The company’s approach is based on defined and proven processes, continual communication and cross-functional collaboration throughout each of the product lifecycle stages.
By allowing companies to work with one experienced partner, KMC 360 smoothes product transitions from design to manufacture, avoiding costly redesigns and delays. It also enhances field maintenance support by allowing end users to leverage KMC Systems’ intimate design knowledge of their systems gained through the design and manufacturing stages.
KMC Systems’ integrated process is ideal for in-vitro diagnostic, surgical, therapeutic and lab automation applications. The company uses its multidisciplinary expertise and 30 years of experience to produce and maintain medical systems such as clinical chemistry analyzers, surgical irrigation pumps, hemodialysis instruments and therapeutic cell processors.
“Relying on multiple partners throughout a product’s lifecycle can result in market launch delays, increased costs and inefficiencies,” says Patrick W. McNallen, president, KMC Systems. “Just as a circle is all encompassing, so too is KMC Systems’ portfolio of services. Our customers work with one partner that has the relevant experience and skill set to develop, produce and maintain complex medical devices and instrumentation.”
KMC 360 helps medical companies bring products to market on time and on budget as well as reduce manufacturing and field service costs, which is especially critical in times of economic difficulty. The company’s approach is based on defined and proven processes, continual communication and cross-functional collaboration throughout each of the product lifecycle stages.
By allowing companies to work with one experienced partner, KMC 360 smoothes product transitions from design to manufacture, avoiding costly redesigns and delays. It also enhances field maintenance support by allowing end users to leverage KMC Systems’ intimate design knowledge of their systems gained through the design and manufacturing stages.
KMC Systems’ integrated process is ideal for in-vitro diagnostic, surgical, therapeutic and lab automation applications. The company uses its multidisciplinary expertise and 30 years of experience to produce and maintain medical systems such as clinical chemistry analyzers, surgical irrigation pumps, hemodialysis instruments and therapeutic cell processors.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!