A Paradigm Shift to a Culture of Quality
Make this shift before your competitors do.

Manufacturing professionals in quality assurance and process improvement are not new to understanding the importance of quality. However, do they know the difference to be made in creating a culture of quality with regards to driving the policies, practices, and processes needed to accomplish an organization’s work? Developing a culture of quality begins with embodying core values: guiding philosophies, behaviors, and attitudes that, when combined, contribute to day-to-day operations. The culture of quality matures over the decades, as norms are passed from one generation of employees to the next. To transition an organization’s culture to embody quality, it requires commitment and deliberate management of the change processes.
As quality leaders, it’s our role to drive differentiation for our companies. It starts with quality process engagement across functions and ensures that quality is a high priority with top management. Strategy, data and analysis can help communicate this to top management in order to drive a change in culture.
ISO 9001:2015
ISO 9001, the international standard that specifies requirements for a quality management system (QMS) helps organizations to consistently provide products that meet regulatory requirements. How does the ISO revision, ISO 9001:2015, come into play in building a culture of quality? It makes top management responsible and accountable for carrying out a certain number of activities themselves. ISO 9001:2015 helps bring together people at the highest levels into the quality management system. No longer does it center on the quality management department. Leadership must promote quality management as a process and understand how all processes fit together.
Quality Change Agents
We need change agents. This is where we need to wear a lot of hats, be business-savvy and emphasize quality. A great supporting tag line is “quality starts with me.” Many organizations use similar tag lines to ensure everyone understands that they play an important role in adopting a culture of quality. It helps cross-functional leaders, teams and individual contributors to understand their role within the organization and how it impacts the final product and perception by customers. This is a good place to cultivate where quality initiatives can begin. Embody that tag line and lead the journey to transform quality away from just a conformance activity to a performance activity.
Quality experts need to make that shift first, and the help to lead the shift across the company so that the right results are achieved—meaning, everything comes back to quality. Over time we have seen that quality is becoming a more important element in differentiating a company in this increasingly difficult time. ISO 9001: 2015 adds to the competitive landscape. Make this shift before your competitors do.
The Digital Age: Quality and Innovation
According to KMPG, digital transformation is the application of digital technologies to fundamentally impact all aspects of business and society. This transformation brings increased quality and innovation. To this point, LNS Research performed a study in which 1,200 executives from various industries were benchmarked. The study showed that the digital transformation and the Internet of Things (IoT) is driving quality improvement today and will continue to do so the future.
The digital transformation requires that same evolution in our approach to business processes. Better to spend less time bickering about who will do what and whose turf it is, and invest time on carrying out a digital transformation. This is how to achieve operational excellence, define a framework to support the processes and build business cases for change. The final step is choosing the technology to make the digital transformation real.
Quality and Operational Excellence
What is an effective quality management strategy? It’s a multi-year plan that aligns quality with strategic objectives through improvements across operational excellence—people, process and technology. There is an “operational architecture” that takes into account the IT infrastructure, with such systems as PLM (product lifecycle management) and ERP (enterprise resource planning), from an IT perspective as well as the “operational technology” on the shop floor and with in-service equipment. The strategic objective looks across operational excellence and operational architectures and delivers value to the business.
Use a transformation framework that helps define the strategies. There can be several initiatives going on at the same time. There are process, culture, and technology initiatives that are occurring at different levels of maturity but that still all fit into a framework.
Realigning People, Process and Technology
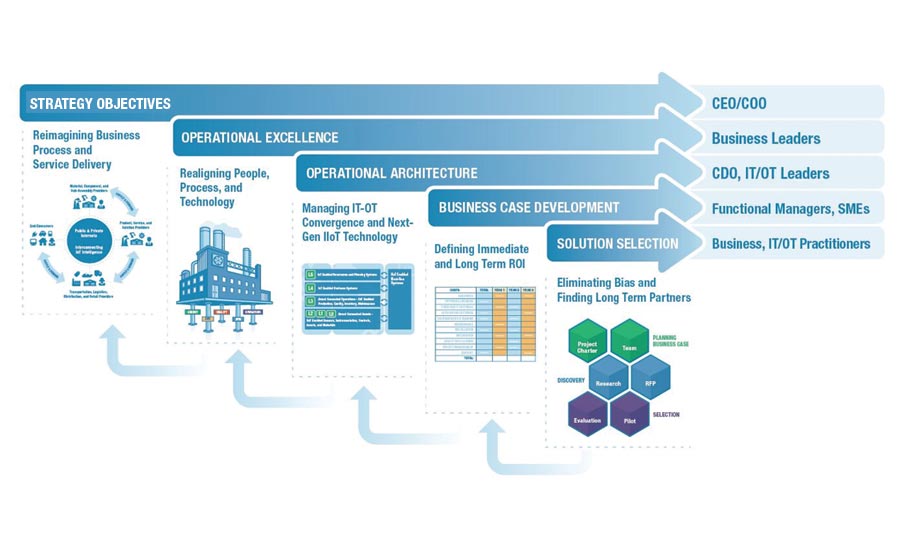
In building a strategy framework, instill a plan of action or policy designed to achieve a major goal. First off, the strategic objectives are set by the C-level, where the organization re-imagines business process and service delivery. Next, operational excellence is upheld by the business leaders within the organization that aim to realign people, process and technology.
In building the plan, functional managers must devise business case development to define immediate and long-term ROI. Once this is achieved, the solution can be selected by IT/OT practitioners, based all on the criteria developed and hopefully eliminating bias, while also seeking long-term partners. Put in culture-building activities. It’s a pressure-packed scenario because the rules are defined around the drive to quality, but that does not actually have anything to do with compliance.
Need for Improvement
While it’s important to be compliant, conformance isn’t necessarily the strategy to become compliant. Conformance doesn’t gain top management support, build culture, drive operational excellence or meet performance objectives. In fact, LNS research shows that 92 percent of quality processes are not harmonized at the average-sized manufacturer. Almost 87 percent of companies do not have top management priority for quality. Approximately 77 percent of manufacturers do not have a culture of quality and 37 percent of manufacturers can’t meet objectives due to ineffective metrics.
Core Principles
So how does ISO 9001: 2015 tie into this framework? First of all, a positive or negative event often can occur, driving a business influence or pressure that forces action and response with a defined date and business owner. For instance, a manufacturer of biosensors and blood glucose monitors sought to create a safe, high-quality medical device for use worldwide, and that meant ensuring all country-specific and local standards would be met. The driving factor caused them to work with several external auditors and legal counselors to ensure compliance was achieved in every jurisdiction. PLM software helped them to ensure all regulations were met and tracked throughout the product lifecycle of their medical device. It properly tracked all electronic signatures, providing electronic audit trails/history tracking, security controls and reporting.
The PLM system supported ISO 9001 requirements and made the audit process streamlined. This is an event that drove a business response because it required someone in top management to own the follow-through of an action that directly influenced the business. For this manufacturer, ISO 9001 ties in well to this scenario. Any potential crisis in terms of achieving compliance and being fined was averted. Too often, quality gets executive priority on the heels of some negative event or crisis that occurs to cause change. It’s far better to get in front of that and make changes to combat challenges prior to a crisis situation. Why not implement a quality initiative when everything is going well?
Building Bridges – Getting Consensus
Get executive sponsorship and management buy-in. Find out what the executive management team measures in terms of their KPIs (key performance indicators) and how effectively they are achieving key business objectives. Align your quality initiative to address the objectives of executive management, as that will help determine their buy-in to follow through on achieving the quality initiatives presented.
Companies can have all kinds of challenges when it comes to quality, such as cultural issues. Those in charge of quality can be seen as the police, preventers of defects, and may not be seen in the best light. This is a typical cultural challenge that would need to be addressed. ISO 9001: 2015 can help address such issues.
A Quality Management Maturity Model
LNS’s research uncovered five levels that characterize a quality management maturity model. It’s not a process model but rather an operational excellence model. Starting at the top are innovation leaders who drive the standards and expectations. Under that are the agile companies comprising evolved people, processes and technology. Next comes the harmonized level, which embraces flexibility unified across the organization; and below that is the controlled level, which is repeatable within organizational, process and/or technology boundaries. At the lowest echelon is the ad hoc unstandardized level displaying significant variation.
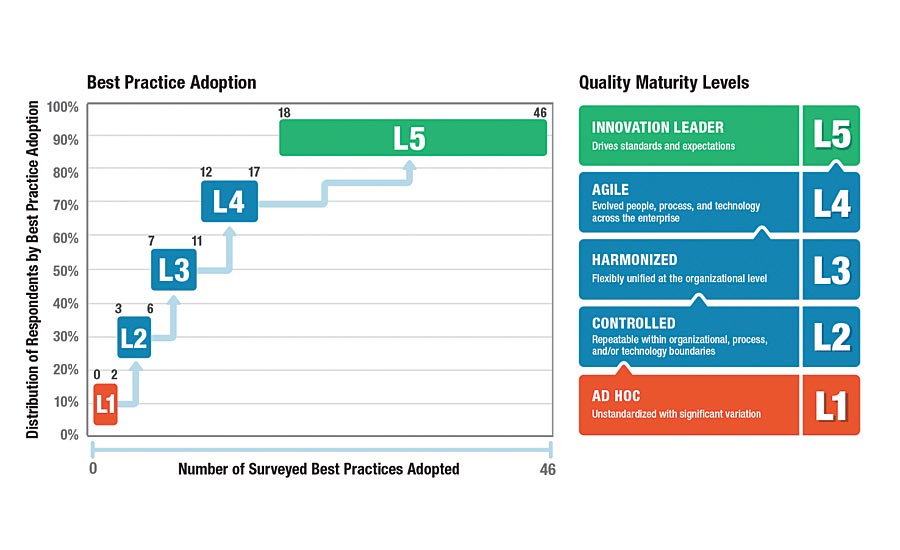
There is a direct correlation between the level at which an organization is operating and whether they have a top-down approach versus a middle-up one. There are numerous benchmarks and research studies to support this. Top management sponsorship matters. Those companies that have top management sponsorship have 2.6 percent more best practices adoption than those that don’t. There needs to be a shift from being a cost center to a value center.
Key Challenges
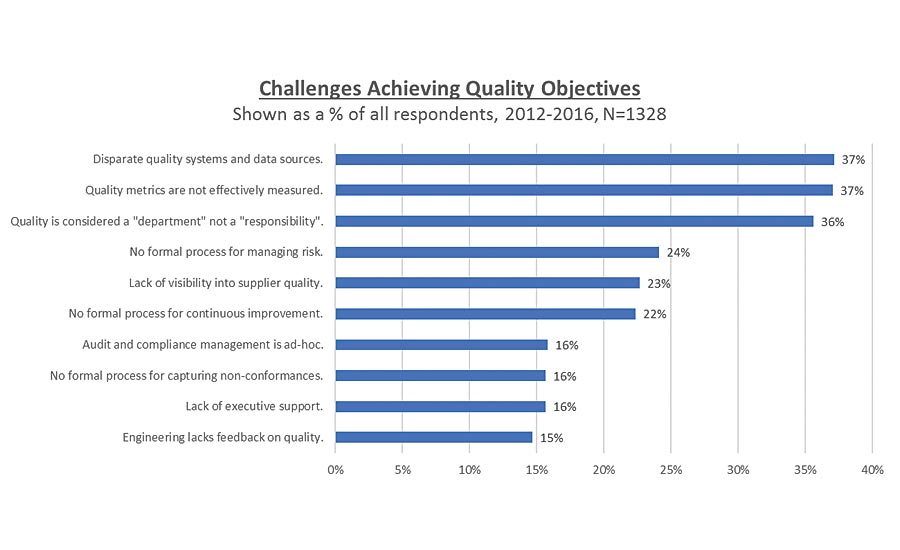
Core management principles of ISO 9001: 2015 can help address challenges for avoiding nonconformities during any certification audit and can help with the issues that the LNS benchmark study uncovered. For instance, the study revealed that 37 percent of respondents found dealing with disparate quality systems and data sources, as well as having quality metrics not effectively measured, to be problematic. Over 35 percent of respondents felt that quality was a department, not a responsibility. Almost 25 percent reported that they had no formal process in place for managing risk. More than 20 percent of all benchmark participants admitted that there was a lack of visibility into supplier quality and no defined process for continual improvement. There were 16 percent who answered that they had no formal process for capturing non-conformances. Overall there was a lack of executive support, and audit/compliance management was dealt with on an ad hoc basis.
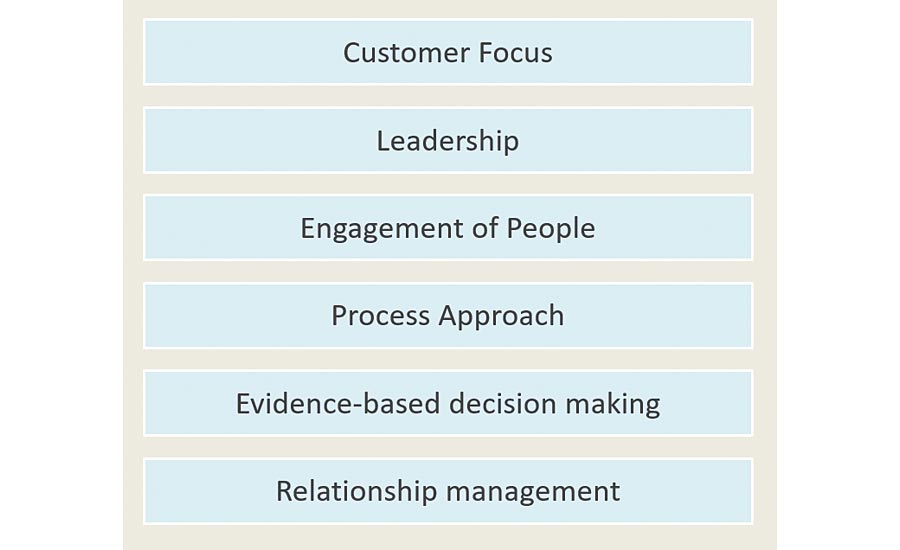
The statistics show gaps where the industry stands in the adoption of the core principles of ISO 9001: 2015, which encompass: customer focus, leadership, engagement of people, process approach, improvement, evidence-based decision-making and relationship management. The benchmark study showed 89 percent of SMEs do not have visibility into real-time quality metrics across functions and some 87 percent do not have top management executive buy-in for quality. With regard to the engagement of people, about 77 percent felt that they do not have a culture of quality adopted. Almost 80 percent did not close the loop on quality processes across design, manufacturing and suppliers. Over 60 percent do not have any visibility into customer complaints and 55 percent haven’t established corrective and preventive action (CAPA) processes across the company.
PLM software can be implemented to assist with quality and support quality management. For instance, EST Analytical adopted a single engineering change and quality control PLM system to help meet ISO compliance. In EST Analytical’s case, they lacked control over their information because it was not indexed or linked. Using this PLM-based quality system, they created a more efficient and connected environment that would easily manage and track information and simplify data sharing across the company. The engineering/R&D, manufacturing, purchasing, quality, sales and service departments use PLM for change management, bill of material (BOM) management, quality management, training management and project management to integrate various manual processes into a single automated system with traceability on all changes.
Best Practices
Understand your “as is” state and then define your “to be” state. It’s a multi-step operation that takes time and effort. It requires process and technology changes. Increase your maturity in your quest for quality continually and gather metrics to work towards this goal. Try to capture metrics around the cost of poor quality. Figure out a way to capture and use the data to deliver a real metric. LNS’s benchmark addressed 46 best practices and then mapped the statistics to their quality maturity model to determine the key areas that a company should focus on.
This research uncovered the best practices to reach ISO 9005:2015 conformance. Adopting these processes and systems also help to reach the ultimate goal of better performance. Highlighted here are a few of the best practices that touch on conformance and performance:
- Develop closed loop processes established to connect quality across design, manufacturers and suppliers.
- Implement compliance processes that are easily understood and documented
- Build cross-functional teams to manage quality across design, manufacturers and suppliers.
- Implement Enterprise Quality Management Software (EQMS)
- Gain real-time visibility of quality metrics in customer service, engineering and manufacturing
- Identify, qualify, and prioritize risk factors across operations
- Ensure formal audit management processes are established to understand current compliance status and identify areas for improvement
- Automate risk management with software, as well as track and visualize risk factors across operations
- Establish the ability to share compliance processes across functional areas
- Make quality an executive top priority
Incorporate the actions outlined in the above bullet points—the impact is tremendous. Successful organizations use ISO 9001: 2015 to drive top management’s priority to quality. It truly will help SMEs achieve operational excellence. Shift from conformance to performance and, in the end, you’ll gain a critical competitive advantage.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!




